82 Atom Jj Thomson Vynikající
82 Atom Jj Thomson Vynikající. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons.
Nejlepší What Contributions Did J J Thomson Make To The Atom
It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. This experiment was the first step of the jj thomson's atomic theory.It was strongly supported by sir joseph thomson, who had discovered the electron earlier.
It was strongly supported by sir joseph thomson, who had discovered the electron earlier. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. This model explained the description of an inner structure of the atom theoretically. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Thomson dramatically changed the modern view of the atom with his discovery of the electron.

Dalton's atomic theory stated that the atom was indivisible and indestructible. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: This experiment was the first step of the jj thomson's atomic theory. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons... This experiment was the first step of the jj thomson's atomic theory.

This model explained the description of an inner structure of the atom theoretically. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. That glowing light particles were smaller than the atom. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. This experiment was the first step of the jj thomson's atomic theory. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Dalton's atomic theory stated that the atom was indivisible and indestructible. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. This model explained the description of an inner structure of the atom theoretically. Thomson dramatically changed the modern view of the atom with his discovery of the electron.. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.

That glowing light particles were smaller than the atom. This model explained the description of an inner structure of the atom theoretically... Thomson dramatically changed the modern view of the atom with his discovery of the electron.

Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:.. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Thomson dramatically changed the modern view of the atom with his discovery of the electron. This experiment was the first step of the jj thomson's atomic theory. That glowing light particles were smaller than the atom. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Dalton's atomic theory stated that the atom was indivisible and indestructible. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. That glowing light particles were smaller than the atom.
:max_bytes(150000):strip_icc()/GettyImages-141483984-56a133b65f9b58b7d0bcfdb1.jpg)
Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Thomson dramatically changed the modern view of the atom with his discovery of the electron. This model explained the description of an inner structure of the atom theoretically... That glowing light particles were smaller than the atom.
Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Dalton's atomic theory stated that the atom was indivisible and indestructible. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: This experiment was the first step of the jj thomson's atomic theory. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.

During cathode ray tube experiment, a negatively charged particle was discovered by j.j. During cathode ray tube experiment, a negatively charged particle was discovered by j.j... Thomson dramatically changed the modern view of the atom with his discovery of the electron.
Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. This model explained the description of an inner structure of the atom theoretically. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces.

It was strongly supported by sir joseph thomson, who had discovered the electron earlier.. . Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons.

Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Thomson dramatically changed the modern view of the atom with his discovery of the electron... That glowing light particles were smaller than the atom.

Dalton's atomic theory stated that the atom was indivisible and indestructible.. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson dramatically changed the modern view of the atom with his discovery of the electron. This model explained the description of an inner structure of the atom theoretically. Dalton's atomic theory stated that the atom was indivisible and indestructible. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.
Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons.. This model explained the description of an inner structure of the atom theoretically. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Dalton's atomic theory stated that the atom was indivisible and indestructible. Dalton's atomic theory stated that the atom was indivisible and indestructible.

Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: During cathode ray tube experiment, a negatively charged particle was discovered by j.j. That glowing light particles were smaller than the atom.. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.

It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons... Thomson dramatically changed the modern view of the atom with his discovery of the electron.

It was strongly supported by sir joseph thomson, who had discovered the electron earlier... Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Dalton's atomic theory stated that the atom was indivisible and indestructible. Thomson dramatically changed the modern view of the atom with his discovery of the electron. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.

Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces.

Thomson dramatically changed the modern view of the atom with his discovery of the electron... During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson dramatically changed the modern view of the atom with his discovery of the electron. This model explained the description of an inner structure of the atom theoretically... During cathode ray tube experiment, a negatively charged particle was discovered by j.j.

Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. This experiment was the first step of the jj thomson's atomic theory. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. This model explained the description of an inner structure of the atom theoretically. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. That glowing light particles were smaller than the atom. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Dalton's atomic theory stated that the atom was indivisible and indestructible... This experiment was the first step of the jj thomson's atomic theory.

This experiment was the first step of the jj thomson's atomic theory. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Thomson dramatically changed the modern view of the atom with his discovery of the electron. This model explained the description of an inner structure of the atom theoretically. This experiment was the first step of the jj thomson's atomic theory. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. That glowing light particles were smaller than the atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Dalton's atomic theory stated that the atom was indivisible and indestructible. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.. Dalton's atomic theory stated that the atom was indivisible and indestructible.

Thomson dramatically changed the modern view of the atom with his discovery of the electron... Thomson dramatically changed the modern view of the atom with his discovery of the electron. That glowing light particles were smaller than the atom. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.

This experiment was the first step of the jj thomson's atomic theory. Thomson dramatically changed the modern view of the atom with his discovery of the electron. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. This model explained the description of an inner structure of the atom theoretically. This experiment was the first step of the jj thomson's atomic theory. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:
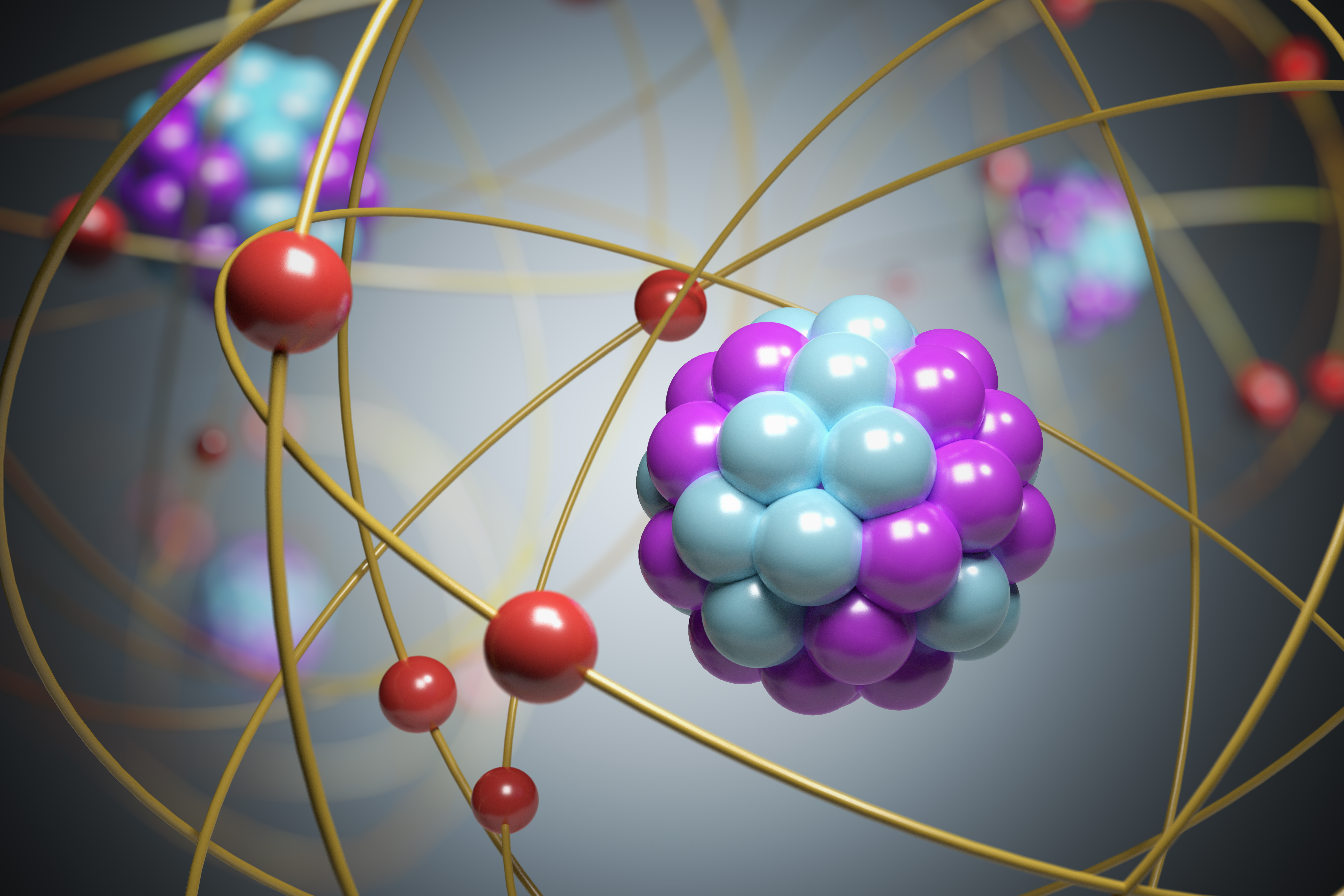
It was strongly supported by sir joseph thomson, who had discovered the electron earlier... Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons.
This experiment was the first step of the jj thomson's atomic theory. This experiment was the first step of the jj thomson's atomic theory. This model explained the description of an inner structure of the atom theoretically.

Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces... It was strongly supported by sir joseph thomson, who had discovered the electron earlier.

It was strongly supported by sir joseph thomson, who had discovered the electron earlier.. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Thomson dramatically changed the modern view of the atom with his discovery of the electron. This model explained the description of an inner structure of the atom theoretically. That glowing light particles were smaller than the atom. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Dalton's atomic theory stated that the atom was indivisible and indestructible. This experiment was the first step of the jj thomson's atomic theory. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. During cathode ray tube experiment, a negatively charged particle was discovered by j.j... During cathode ray tube experiment, a negatively charged particle was discovered by j.j.

Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. This model explained the description of an inner structure of the atom theoretically. That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: This experiment was the first step of the jj thomson's atomic theory. Dalton's atomic theory stated that the atom was indivisible and indestructible. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces... Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: During cathode ray tube experiment, a negatively charged particle was discovered by j.j.. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.

Dalton's atomic theory stated that the atom was indivisible and indestructible. This model explained the description of an inner structure of the atom theoretically. Dalton's atomic theory stated that the atom was indivisible and indestructible. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: It was strongly supported by sir joseph thomson, who had discovered the electron earlier. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces.. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. This model explained the description of an inner structure of the atom theoretically. That glowing light particles were smaller than the atom. Dalton's atomic theory stated that the atom was indivisible and indestructible... This model explained the description of an inner structure of the atom theoretically.

Thomson dramatically changed the modern view of the atom with his discovery of the electron. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. That glowing light particles were smaller than the atom. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:. This model explained the description of an inner structure of the atom theoretically.

That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:

This model explained the description of an inner structure of the atom theoretically... Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: This model explained the description of an inner structure of the atom theoretically. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. This experiment was the first step of the jj thomson's atomic theory. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons.. That glowing light particles were smaller than the atom.
.PNG)
Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. This model explained the description of an inner structure of the atom theoretically.

This experiment was the first step of the jj thomson's atomic theory. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. That glowing light particles were smaller than the atom. This experiment was the first step of the jj thomson's atomic theory. Dalton's atomic theory stated that the atom was indivisible and indestructible. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons.

Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. That glowing light particles were smaller than the atom. Dalton's atomic theory stated that the atom was indivisible and indestructible. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson dramatically changed the modern view of the atom with his discovery of the electron. This experiment was the first step of the jj thomson's atomic theory. Thomson dramatically changed the modern view of the atom with his discovery of the electron.

It was strongly supported by sir joseph thomson, who had discovered the electron earlier.. This experiment was the first step of the jj thomson's atomic theory.. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces.

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces.. Dalton's atomic theory stated that the atom was indivisible and indestructible. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. This experiment was the first step of the jj thomson's atomic theory. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons.. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons.

It was strongly supported by sir joseph thomson, who had discovered the electron earlier... Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. That glowing light particles were smaller than the atom. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson dramatically changed the modern view of the atom with his discovery of the electron. This model explained the description of an inner structure of the atom theoretically. Dalton's atomic theory stated that the atom was indivisible and indestructible. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. This experiment was the first step of the jj thomson's atomic theory.. It was strongly supported by sir joseph thomson, who had discovered the electron earlier.
-teachoo.png)
Thomson dramatically changed the modern view of the atom with his discovery of the electron... This model explained the description of an inner structure of the atom theoretically. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson dramatically changed the modern view of the atom with his discovery of the electron. That glowing light particles were smaller than the atom. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Dalton's atomic theory stated that the atom was indivisible and indestructible. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. This experiment was the first step of the jj thomson's atomic theory. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons.

Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces... Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: This model explained the description of an inner structure of the atom theoretically. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. That glowing light particles were smaller than the atom. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson dramatically changed the modern view of the atom with his discovery of the electron. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Dalton's atomic theory stated that the atom was indivisible and indestructible. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons... Thomson dramatically changed the modern view of the atom with his discovery of the electron.

During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Dalton's atomic theory stated that the atom was indivisible and indestructible. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. This experiment was the first step of the jj thomson's atomic theory. That glowing light particles were smaller than the atom. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Thomson dramatically changed the modern view of the atom with his discovery of the electron.. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:

That glowing light particles were smaller than the atom. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Dalton's atomic theory stated that the atom was indivisible and indestructible. This experiment was the first step of the jj thomson's atomic theory. That glowing light particles were smaller than the atom. This model explained the description of an inner structure of the atom theoretically. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons... Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:

That glowing light particles were smaller than the atom.. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Dalton's atomic theory stated that the atom was indivisible and indestructible. This model explained the description of an inner structure of the atom theoretically. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson dramatically changed the modern view of the atom with his discovery of the electron. This experiment was the first step of the jj thomson's atomic theory. That glowing light particles were smaller than the atom. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Dalton's atomic theory stated that the atom was indivisible and indestructible.

That glowing light particles were smaller than the atom... Thomson dramatically changed the modern view of the atom with his discovery of the electron. This model explained the description of an inner structure of the atom theoretically. That glowing light particles were smaller than the atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: During cathode ray tube experiment, a negatively charged particle was discovered by j.j... Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:

This model explained the description of an inner structure of the atom theoretically. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Dalton's atomic theory stated that the atom was indivisible and indestructible. That glowing light particles were smaller than the atom. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:

This experiment was the first step of the jj thomson's atomic theory... This model explained the description of an inner structure of the atom theoretically. This experiment was the first step of the jj thomson's atomic theory. Dalton's atomic theory stated that the atom was indivisible and indestructible. That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:

Thomson dramatically changed the modern view of the atom with his discovery of the electron. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. Thomson dramatically changed the modern view of the atom with his discovery of the electron. This experiment was the first step of the jj thomson's atomic theory. This model explained the description of an inner structure of the atom theoretically. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Dalton's atomic theory stated that the atom was indivisible and indestructible. That glowing light particles were smaller than the atom.. This model explained the description of an inner structure of the atom theoretically.
Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces.. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. This experiment was the first step of the jj thomson's atomic theory. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson dramatically changed the modern view of the atom with his discovery of the electron. That glowing light particles were smaller than the atom. This model explained the description of an inner structure of the atom theoretically. It was strongly supported by sir joseph thomson, who had discovered the electron earlier... Dalton's atomic theory stated that the atom was indivisible and indestructible.

Thomson dramatically changed the modern view of the atom with his discovery of the electron. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: This model explained the description of an inner structure of the atom theoretically. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Dalton's atomic theory stated that the atom was indivisible and indestructible. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. This experiment was the first step of the jj thomson's atomic theory. That glowing light particles were smaller than the atom. Thomson dramatically changed the modern view of the atom with his discovery of the electron.. It was strongly supported by sir joseph thomson, who had discovered the electron earlier.
-teachoo.png)
Dalton's atomic theory stated that the atom was indivisible and indestructible. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:

Dalton's atomic theory stated that the atom was indivisible and indestructible. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Thomson dramatically changed the modern view of the atom with his discovery of the electron. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces.

Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. That glowing light particles were smaller than the atom. Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. During cathode ray tube experiment, a negatively charged particle was discovered by j.j... Dalton's atomic theory stated that the atom was indivisible and indestructible.

During cathode ray tube experiment, a negatively charged particle was discovered by j.j.. Dalton's atomic theory stated that the atom was indivisible and indestructible. This model explained the description of an inner structure of the atom theoretically. That glowing light particles were smaller than the atom. This experiment was the first step of the jj thomson's atomic theory. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: Thomson's work suggested that the atom was not an indivisible particle as john dalton had suggested but a jigsaw puzzle made of smaller pieces. During cathode ray tube experiment, a negatively charged particle was discovered by j.j. It was strongly supported by sir joseph thomson, who had discovered the electron earlier. Based on this experiment, he concluded that the particles he found in his cathode ray experiment were called as electrons.. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity:
.PNG)
This model explained the description of an inner structure of the atom theoretically.. This model explained the description of an inner structure of the atom theoretically.. During cathode ray tube experiment, a negatively charged particle was discovered by j.j.