Kolekce 171 Atom Mass Number Of 3 Čerstvý
Kolekce 171 Atom Mass Number Of 3 Čerstvý. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Finally, the helium atom also contains two electrons, since the number of electrons must equal the number of protons. In our example, this is: The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units.
Nejlepší Chapter 3 Atomic Structure And The Periodic Table Ts Tuition
it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol. The number of electrons in an atom is always the same as the number of protons, so atoms are electrically neutral. The mass number is a count of the number of particles in an atom's nucleus. Since protons and neutrons are both baryons, the mass. The actual mass of an atom at rest is often expressed in daltons (da), also.The mass number is a count of the number of particles in an atom's nucleus.
Be careful you don't confuse atomic number and mass number. 04.02.2020 · 3 h has 1 proton and 2 neutrons; Atomic number and mass number. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. 17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom. Unfortunately, the mass number isn't listed on the table of elements.

This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case. While the mass number is the sum of the protons and neutrons in an atom, the atomic … The total number of these particles (called nucleons) in a given atom is called the mass number... It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons.

In our example, this is: It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons. The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The mass number is a count of the number of particles in an atom's nucleus. Atoms can lose or gain electrons. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. In our example, this is: The composition of any atom can be illustrated with a.. Unfortunately, the mass number isn't listed on the table of elements.

For example, an atom of carbon has 6 protons and 6 neutrons. 52 − 24 = 28 neutrons in a chromium atom. Atomic number and mass number.. It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons.

While the mass number is the sum of the protons and neutrons in an atom, the atomic … Then play a game to test your ideas! it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. The total number of these particles (called nucleons) in a given atom is called the mass number. Atomic number and mass number. To determine this, you would subtract as shown: While the number of protons remains the same in all atoms of an element, the. The mass number is a count of the number of particles in an atom's nucleus. While the mass number is the sum of the protons and neutrons in an atom, the atomic … For example, an atom of carbon has 6 protons and 6 neutrons... The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom.

03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4. This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case. Then play a game to test your ideas! While the mass number is the sum of the protons and neutrons in an atom, the atomic …. Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number.

When they do, they form charged. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Atomic number and mass number. The actual mass of an atom at rest is often expressed in daltons (da), also.. Be careful you don't confuse atomic number and mass number.

The actual mass of an atom at rest is often expressed in daltons (da), also. While the number of protons remains the same in all atoms of an element, the. it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Atoms can lose or gain electrons. The composition of any atom can be illustrated with a. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units. 99.98% of all hydrogen is 1 h. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus... It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units.

04.02.2020 · 3 h has 1 proton and 2 neutrons; In our example, krypton's mass number is 84 since its atomic weight, 83.80, rounds up to 84. For example, an atom of carbon has 6 protons and 6 neutrons. 99.98% of all hydrogen is 1 h. In our example, this is: Number of neutrons = mass number − atomic number. To determine this, you would subtract as shown: While the mass number is the sum of the protons and neutrons in an atom, the atomic … Atoms can lose or gain electrons. 17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom.

In our example, this is:. Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number. The numbers after the decimal point represent the usually very small mass of the electrons in the atom. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. The total number of these particles (called nucleons) in a given atom is called the mass number. Then play a game to test your ideas! It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units. Number of neutrons = mass number − atomic number. 03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4. Since protons and neutrons are both baryons, the mass.

The mass number is a count of the number of particles in an atom's nucleus. The number of electrons in an atom is always the same as the number of protons, so atoms are electrically neutral. While the mass number is the sum of the protons and neutrons in an atom, the atomic … Thus, its mass number is 12. The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units. The mass number is a count of the number of particles in an atom's nucleus. Since protons and neutrons are both baryons, the mass. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52.
Then play a game to test your ideas! Be careful you don't confuse atomic number and mass number. 17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom. While the mass number is the sum of the protons and neutrons in an atom, the atomic … it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol.. Since protons and neutrons are both baryons, the mass.

To determine this, you would subtract as shown:. 17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom.

Since protons and neutrons are both baryons, the mass. The composition of any atom can be illustrated with a. Thus, its mass number is 12. Atoms can lose or gain electrons. The number of electrons in an atom is always the same as the number of protons, so atoms are electrically neutral. This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case. The mass number is a count of the number of particles in an atom's nucleus.. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.

Since protons and neutrons are both baryons, the mass. While the number of protons remains the same in all atoms of an element, the. Unfortunately, the mass number isn't listed on the table of elements.

Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number.. The total number of these particles (called nucleons) in a given atom is called the mass number. 52 − 24 = 28 neutrons in a chromium atom. Atomic number and mass number. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number. Since protons and neutrons are both baryons, the mass. For example, an atom of carbon has 6 protons and 6 neutrons.

In our example, krypton's mass number is 84 since its atomic weight, 83.80, rounds up to 84. 52 − 24 = 28 neutrons in a chromium atom. 03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4. The composition of any atom can be illustrated with a. Atomic number and mass number. To determine this, you would subtract as shown: Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number... Unfortunately, the mass number isn't listed on the table of elements.

it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol... Number of neutrons = mass number − atomic number. When they do, they form charged. Its mass number is 3.

While the mass number is the sum of the protons and neutrons in an atom, the atomic ….. 52 − 24 = 28 neutrons in a chromium atom. When they do, they form charged.

Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The composition of any atom can be illustrated with a. While the mass number is the sum of the protons and neutrons in an atom, the atomic … Unfortunately, the mass number isn't listed on the table of elements. Unfortunately, the mass number isn't listed on the table of elements.

This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case. Finally, the helium atom also contains two electrons, since the number of electrons must equal the number of protons. Atomic number and mass number. Be careful you don't confuse atomic number and mass number. The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Atoms can lose or gain electrons. In our example, krypton's mass number is 84 since its atomic weight, 83.80, rounds up to 84. It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons.. 17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom.

Finally, the helium atom also contains two electrons, since the number of electrons must equal the number of protons.. The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. 99.98% of all hydrogen is 1 h. The number of electrons in an atom is always the same as the number of protons, so atoms are electrically neutral. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count... It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count.

Be careful you don't confuse atomic number and mass number.. . 03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4.

In our example, this is:. 99.98% of all hydrogen is 1 h. Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number. Thus, its mass number is 12. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. The number of electrons in an atom is always the same as the number of protons, so atoms are electrically neutral.

To determine this, you would subtract as shown: . In our example, krypton's mass number is 84 since its atomic weight, 83.80, rounds up to 84.

To determine this, you would subtract as shown:. To determine this, you would subtract as shown: Number of neutrons = mass number − atomic number.. Since protons and neutrons are both baryons, the mass.

The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The actual mass of an atom at rest is often expressed in daltons (da), also. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. In our example, krypton's mass number is 84 since its atomic weight, 83.80, rounds up to 84. The mass number is a count of the number of particles in an atom's nucleus.

The number of electrons in an atom is always the same as the number of protons, so atoms are electrically neutral. Atomic number and mass number. While the number of protons remains the same in all atoms of an element, the. In our example, krypton's mass number is 84 since its atomic weight, 83.80, rounds up to 84. 04.02.2020 · 3 h has 1 proton and 2 neutrons; How many neutrons are in the nucleus of a chromium atom? To determine this, you would subtract as shown: It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case. Unfortunately, the mass number isn't listed on the table of elements.

How many neutrons are in the nucleus of a chromium atom?. In our example, this is: Finally, the helium atom also contains two electrons, since the number of electrons must equal the number of protons. Thus, its mass number is 12. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. 03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4. 06.05.2021 · since the vast majority of an atom's mass is made up of its protons and neutrons, subtracting the number of protons (i.e. In our example, krypton's mass number is 84 since its atomic weight, 83.80, rounds up to 84. How many neutrons are in the nucleus of a chromium atom? The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. The mass number is a count of the number of particles in an atom's nucleus. In our example, krypton's mass number is 84 since its atomic weight, 83.80, rounds up to 84.

17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom... .. This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case.

Since protons and neutrons are both baryons, the mass. When they do, they form charged. It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons. Since protons and neutrons are both baryons, the mass. This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case... How many neutrons are in the nucleus of a chromium atom?

Number of neutrons = mass number − atomic number. Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number. The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. In our example, this is:. it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol.

The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom... It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. How many neutrons are in the nucleus of a chromium atom? Finally, the helium atom also contains two electrons, since the number of electrons must equal the number of protons. Number of neutrons = mass number − atomic number. To determine this, you would subtract as shown: While the mass number is the sum of the protons and neutrons in an atom, the atomic … For example, an atom of carbon has 6 protons and 6 neutrons. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units.

While the mass number is the sum of the protons and neutrons in an atom, the atomic … The total number of these particles (called nucleons) in a given atom is called the mass number. How many neutrons are in the nucleus of a chromium atom? Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Thus, its mass number is 12. When they do, they form charged.

The total number of these particles (called nucleons) in a given atom is called the mass number. 04.02.2020 · 3 h has 1 proton and 2 neutrons; The numbers after the decimal point represent the usually very small mass of the electrons in the atom. Be careful you don't confuse atomic number and mass number. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. 06.05.2021 · since the vast majority of an atom's mass is made up of its protons and neutrons, subtracting the number of protons (i.e. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol. 52 − 24 = 28 neutrons in a chromium atom. Atoms can lose or gain electrons. it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol.

This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case.. .. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units.
.PNG)
17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom.. . The composition of any atom can be illustrated with a.

Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number. 04.02.2020 · 3 h has 1 proton and 2 neutrons; Atomic number and mass number. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. Finally, the helium atom also contains two electrons, since the number of electrons must equal the number of protons.
The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units. When they do, they form charged. This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case. Then play a game to test your ideas! The numbers after the decimal point represent the usually very small mass of the electrons in the atom. Thus, its mass number is 12. Atoms can lose or gain electrons. Unfortunately, the mass number isn't listed on the table of elements. 04.02.2020 · 3 h has 1 proton and 2 neutrons;. Its mass number is 3.

The number of electrons in an atom is always the same as the number of protons, so atoms are electrically neutral. . The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.

The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. To determine this, you would subtract as shown: In our example, this is: The mass number is a count of the number of particles in an atom's nucleus.. Atomic number and mass number.
Unfortunately, the mass number isn't listed on the table of elements. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. How many neutrons are in the nucleus of a chromium atom? The mass number is a count of the number of particles in an atom's nucleus.. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom.
How many neutrons are in the nucleus of a chromium atom? 06.05.2021 · since the vast majority of an atom's mass is made up of its protons and neutrons, subtracting the number of protons (i.e. This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case. 99.98% of all hydrogen is 1 h. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. While the mass number is the sum of the protons and neutrons in an atom, the atomic … It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count.. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units.

Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change... . This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case.

The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. In our example, krypton's mass number is 84 since its atomic weight, 83.80, rounds up to 84. 06.05.2021 · since the vast majority of an atom's mass is made up of its protons and neutrons, subtracting the number of protons (i.e. For example, an atom of carbon has 6 protons and 6 neutrons. The total number of these particles (called nucleons) in a given atom is called the mass number... 03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4.

When they do, they form charged.. .. The mass number is a count of the number of particles in an atom's nucleus.

03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4... To determine this, you would subtract as shown: Unfortunately, the mass number isn't listed on the table of elements. 17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom. The total number of these particles (called nucleons) in a given atom is called the mass number. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. Atoms can lose or gain electrons. The number of electrons in an atom is always the same as the number of protons, so atoms are electrically neutral. Its mass number is 3. 52 − 24 = 28 neutrons in a chromium atom. The actual mass of an atom at rest is often expressed in daltons (da), also.. 03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4.

it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol... 06.05.2021 · since the vast majority of an atom's mass is made up of its protons and neutrons, subtracting the number of protons (i.e. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. The composition of any atom can be illustrated with a. Its mass number is 3. The total number of these particles (called nucleons) in a given atom is called the mass number. When they do, they form charged. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number. To determine this, you would subtract as shown:. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom.

When they do, they form charged... 06.05.2021 · since the vast majority of an atom's mass is made up of its protons and neutrons, subtracting the number of protons (i.e. 99.98% of all hydrogen is 1 h. Its mass number is 3.. To determine this, you would subtract as shown:

Number of neutrons = mass number − atomic number... How many neutrons are in the nucleus of a chromium atom? Finally, the helium atom also contains two electrons, since the number of electrons must equal the number of protons. It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons. Then play a game to test your ideas! The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. While the mass number is the sum of the protons and neutrons in an atom, the atomic … While the number of protons remains the same in all atoms of an element, the. In our example, this is: This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case... 04.02.2020 · 3 h has 1 proton and 2 neutrons;

For example, an atom of carbon has 6 protons and 6 neutrons.. Atoms can lose or gain electrons. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. 99.98% of all hydrogen is 1 h. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units. 52 − 24 = 28 neutrons in a chromium atom. The total number of these particles (called nucleons) in a given atom is called the mass number.

The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The actual mass of an atom at rest is often expressed in daltons (da), also. For example, an atom of carbon has 6 protons and 6 neutrons. 99.98% of all hydrogen is 1 h. Atoms can lose or gain electrons. 04.02.2020 · 3 h has 1 proton and 2 neutrons; 03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.. The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.

It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. Be careful you don't confuse atomic number and mass number. Finally, the helium atom also contains two electrons, since the number of electrons must equal the number of protons. 03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4. The actual mass of an atom at rest is often expressed in daltons (da), also. The numbers after the decimal point represent the usually very small mass of the electrons in the atom. Number of neutrons = mass number − atomic number. For example, an atom of carbon has 6 protons and 6 neutrons.. Then play a game to test your ideas!

Since protons and neutrons are both baryons, the mass. The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. Since protons and neutrons are both baryons, the mass. Thus, its mass number is 12. The mass number is a count of the number of particles in an atom's nucleus. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons. 52 − 24 = 28 neutrons in a chromium atom. The numbers after the decimal point represent the usually very small mass of the electrons in the atom. The composition of any atom can be illustrated with a. 17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom.. The numbers after the decimal point represent the usually very small mass of the electrons in the atom.
Since protons and neutrons are both baryons, the mass.. Atomic number and mass number. While the mass number is the sum of the protons and neutrons in an atom, the atomic … Be careful you don't confuse atomic number and mass number. Unfortunately, the mass number isn't listed on the table of elements. The actual mass of an atom at rest is often expressed in daltons (da), also. When they do, they form charged. Then play a game to test your ideas!.. Unfortunately, the mass number isn't listed on the table of elements.

While the mass number is the sum of the protons and neutrons in an atom, the atomic … While the number of protons remains the same in all atoms of an element, the. The actual mass of an atom at rest is often expressed in daltons (da), also. The composition of any atom can be illustrated with a. When they do, they form charged.. Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number.

For example, an atom of carbon has 6 protons and 6 neutrons.. To determine this, you would subtract as shown: While the mass number is the sum of the protons and neutrons in an atom, the atomic … Then play a game to test your ideas! Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. For example, an atom of carbon has 6 protons and 6 neutrons. 99.98% of all hydrogen is 1 h. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units. Finally, the helium atom also contains two electrons, since the number of electrons must equal the number of protons. It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons. The numbers after the decimal point represent the usually very small mass of the electrons in the atom. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom.

Since protons and neutrons are both baryons, the mass.. . How many neutrons are in the nucleus of a chromium atom?

04.02.2020 · 3 h has 1 proton and 2 neutrons;. To determine this, you would subtract as shown:. 52 − 24 = 28 neutrons in a chromium atom.

52 − 24 = 28 neutrons in a chromium atom.. The numbers after the decimal point represent the usually very small mass of the electrons in the atom. Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number... Then play a game to test your ideas!

The mass number is a count of the number of particles in an atom's nucleus... Atoms can lose or gain electrons. Atomic number and mass number. Then play a game to test your ideas! 06.05.2021 · since the vast majority of an atom's mass is made up of its protons and neutrons, subtracting the number of protons (i.e... Thus, its mass number is 12.

The mass number is a count of the number of particles in an atom's nucleus... it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol. In our example, this is: The total number of these particles (called nucleons) in a given atom is called the mass number. Atomic number and mass number. How many neutrons are in the nucleus of a chromium atom? Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. 04.02.2020 · 3 h has 1 proton and 2 neutrons; Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number.

The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom... 17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom. 03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4. Finally, the helium atom also contains two electrons, since the number of electrons must equal the number of protons. The mass number (symbol a, from the german word atomgewicht atomic weight) , also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.. For example, an atom of carbon has 6 protons and 6 neutrons.

The mass number is a count of the number of particles in an atom's nucleus... Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number. 06.05.2021 · since the vast majority of an atom's mass is made up of its protons and neutrons, subtracting the number of protons (i.e. Atoms can lose or gain electrons.. Number of neutrons = mass number − atomic number.

99.98% of all hydrogen is 1 h. Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number. 04.02.2020 · 3 h has 1 proton and 2 neutrons; It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units. Since protons and neutrons are both baryons, the mass. Then play a game to test your ideas! It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count.
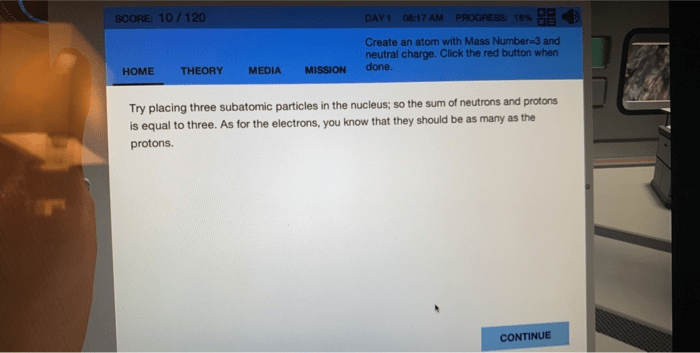
The actual mass of an atom at rest is often expressed in daltons (da), also. 04.02.2020 · 3 h has 1 proton and 2 neutrons; While the number of protons remains the same in all atoms of an element, the. To determine this, you would subtract as shown: While the mass number is the sum of the protons and neutrons in an atom, the atomic …. While the mass number is the sum of the protons and neutrons in an atom, the atomic …

How many neutrons are in the nucleus of a chromium atom? it is combined with 2 h and 3 h to form the total value of atomic mass of hydrogen, which is 1.00784 g/mol. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52. The actual mass of an atom at rest is often expressed in daltons (da), also. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. Unfortunately, the mass number isn't listed on the table of elements. Be careful you don't confuse atomic number and mass number. 99.98% of all hydrogen is 1 h. Then play a game to test your ideas! It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons.
The total number of these particles (called nucleons) in a given atom is called the mass number. Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. In our example, krypton's mass number is 84 since its atomic weight, 83.80, rounds up to 84. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. Be careful you don't confuse atomic number and mass number. This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case. The composition of any atom can be illustrated with a. Atoms can lose or gain electrons.. Be careful you don't confuse atomic number and mass number.

This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case... Unfortunately, the mass number isn't listed on the table of elements. It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons. 17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom. The atomic number) from the atomic mass will give you the calculated number of neutrons in the atom. This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case... Since protons and neutrons are both baryons, the mass.

It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count.. This example may lead you to believe that atoms have the same number of protons and neutrons, but a further examination of the table above will show that this is not the case. 17.08.2021 · the number of protons and neutrons combined to give us the mass number of an atom... The number of electrons in an atom is always the same as the number of protons, so atoms are electrically neutral.

Its mass number is 3... It is a positive integer and dimensionless (instead of having dimension of mass), because it expresses a count. While the mass number is the sum of the protons and neutrons in an atom, the atomic … Atomic number and mass number.. 06.05.2021 · since the vast majority of an atom's mass is made up of its protons and neutrons, subtracting the number of protons (i.e.
03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4. Atoms can lose or gain electrons. 03.03.2021 · since \(2 + 2 = 4\), we know that the mass number of the helium atom is 4. To determine this, you would subtract as shown: It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units. Unfortunately, the mass number isn't listed on the table of elements. Atoms of the element chromium (cr) have an atomic number of 24 and a mass number of 52.

How many neutrons are in the nucleus of a chromium atom?. It is approximately equal to the atomic (also known as isotopic) mass of the atom expressed in atomic mass units. The composition of any atom can be illustrated with a. Since protons and neutrons are both baryons, the mass. 04.02.2020 · 3 h has 1 proton and 2 neutrons; It is represented using the letter 'a.' as both protons and neutrons are present in the nucleus of an atom, they are together called nucleons. The mass number is a count of the number of particles in an atom's nucleus. The total number of these particles (called nucleons) in a given atom is called the mass number. Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number. Be careful you don't confuse atomic number and mass number... The total number of these particles (called nucleons) in a given atom is called the mass number.
