Nápady 81 Carbon Atom Parts
Nápady 81 Carbon Atom Parts. It belongs to group 14 of the periodic table. Carbo coal) is a chemical element with the symbol c and atomic number 6.
Tady Chemical Elements Com Carbon C
Tertiary carbon atoms are branched out in three different ways; Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). 18.03.2008 · what are parts of an atom of carbon? Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. It belongs to group 14 of the periodic table.Carbo coal) is a chemical element with the symbol c and atomic number 6.
It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. But what about the hydrogen atoms which are bonded to these carbon atoms? Quaternary carbon atoms have the most carbon atoms around (max 4). A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); A carbon atom that has four different groups attached is a chiral carbon. Carbon makes up only about 0.025 percent of earth's crust.

It is bonded to three carbon atoms. The internal carbon atom is tertiary (3°);. In this post, we will …

Each of these parts has an associated charge, with protons carrying a positive. In this post, we will … The other three are primary ( …

Carbo coal) is a chemical element with the symbol c and atomic number 6.. Tertiary carbon atoms are branched out in three different ways; It is bonded to three carbon atoms. Atoms consist of three basic particles: But what about the hydrogen atoms which are bonded to these carbon atoms? The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.

Carbo coal) is a chemical element with the symbol c and atomic number 6.. The internal carbon atom is tertiary (3°); 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. It belongs to group 14 of the periodic table. Carbo coal) is a chemical element with the symbol c and atomic number 6. 18.03.2008 · what are parts of an atom of carbon? Carbon makes up only about 0.025 percent of earth's crust. The other three are primary ( …

It belongs to group 14 of the periodic table... It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Carbon makes up only about 0.025 percent of earth's crust. A carbon atom that has four different groups attached is a chiral carbon. 18.03.2008 · what are parts of an atom of carbon? The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Each of these parts has an associated charge, with protons carrying a positive. The internal carbon atom is tertiary (3°); Atoms consist of three basic particles: If a molecule contains one or more chiral carbons, it is likely to ….. Yes, they can also be primary, secondary, and tertiary.

Yes, they can also be primary, secondary, and tertiary.. It is bonded to three carbon atoms. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Carbon makes up only about 0.025 percent of earth's crust. It belongs to group 14 of the periodic table.. Quaternary carbon atoms have the most carbon atoms around (max 4).

Quaternary carbon atoms have the most carbon atoms around (max 4). If a molecule contains one or more chiral carbons, it is likely to … 18.03.2008 · what are parts of an atom of carbon? A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. Yes, they can also be primary, secondary, and tertiary. It belongs to group 14 of the periodic table. The internal carbon atom is tertiary (3°); But what about the hydrogen atoms which are bonded to these carbon atoms? A carbon atom that has four different groups attached is a chiral carbon.. Carbon makes up only about 0.025 percent of earth's crust.

Each of these parts has an associated charge, with protons carrying a positive.. Tertiary carbon atoms are branched out in three different ways; The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Yes, they can also be primary, secondary, and tertiary.. Yes, they can also be primary, secondary, and tertiary.

It belongs to group 14 of the periodic table... It is bonded to three carbon atoms. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. It belongs to group 14 of the periodic table. Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). The internal carbon atom is tertiary (3°);.. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.

The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Carbon makes up only about 0.025 percent of earth's crust. Tertiary carbon atoms are branched out in three different ways; A carbon atom that has four different groups attached is a chiral carbon. Carbo coal) is a chemical element with the symbol c and atomic number 6. The internal carbon atom is tertiary (3°); Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). Atoms consist of three basic particles:.. Tertiary carbon atoms are branched out in three different ways;

Each of these parts has an associated charge, with protons carrying a positive. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); It is bonded to three carbon atoms. 18.03.2008 · what are parts of an atom of carbon?. Each of these parts has an associated charge, with protons carrying a positive.

Carbo coal) is a chemical element with the symbol c and atomic number 6. For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; A carbon atom that has four different groups attached is a chiral carbon. 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Each of these parts has an associated charge, with protons carrying a positive. If a molecule contains one or more chiral carbons, it is likely to … 18.03.2008 · what are parts of an atom of carbon?. If a molecule contains one or more chiral carbons, it is likely to …

A carbon atom that has four different groups attached is a chiral carbon. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; In this post, we will … A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. Carbo coal) is a chemical element with the symbol c and atomic number 6. Carbon makes up only about 0.025 percent of earth's crust. Quaternary carbon atoms have the most carbon atoms around (max 4)... Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom.

But what about the hydrogen atoms which are bonded to these carbon atoms?.. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); Carbo coal) is a chemical element with the symbol c and atomic number 6. It belongs to group 14 of the periodic table. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It is bonded to three carbon atoms. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). 18.03.2008 · what are parts of an atom of carbon? Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. Carbon makes up only about 0.025 percent of earth's crust... Carbon makes up only about 0.025 percent of earth's crust.

19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. Carbon makes up only about 0.025 percent of earth's crust. It is bonded to three carbon atoms... 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);

The other three are primary ( … 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); A carbon atom that has four different groups attached is a chiral carbon. The internal carbon atom is tertiary (3°); The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).. A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.

It is bonded to three carbon atoms. If a molecule contains one or more chiral carbons, it is likely to … In this post, we will … 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements.. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).

The internal carbon atom is tertiary (3°);. A carbon atom that has four different groups attached is a chiral carbon. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. But what about the hydrogen atoms which are bonded to these carbon atoms? The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). The other three are primary ( … A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. It is bonded to three carbon atoms... Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom.

Each of these parts has an associated charge, with protons carrying a positive. 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. Each of these parts has an associated charge, with protons carrying a positive.

The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).. A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.. But what about the hydrogen atoms which are bonded to these carbon atoms?

07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. Each of these parts has an associated charge, with protons carrying a positive. For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary;. In this post, we will …
09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); A carbon atom that has four different groups attached is a chiral carbon.. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°.

It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.. For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; It is bonded to three carbon atoms.

Carbo coal) is a chemical element with the symbol c and atomic number 6. Yes, they can also be primary, secondary, and tertiary. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. Each of these parts has an associated charge, with protons carrying a positive. Carbon makes up only about 0.025 percent of earth's crust. But what about the hydrogen atoms which are bonded to these carbon atoms? Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; Quaternary carbon atoms have the most carbon atoms around (max 4). Atoms consist of three basic particles:.. 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached.
But what about the hydrogen atoms which are bonded to these carbon atoms? 18.03.2008 · what are parts of an atom of carbon? Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. Tertiary carbon atoms are branched out in three different ways; Each of these parts has an associated charge, with protons carrying a positive.
18.03.2008 · what are parts of an atom of carbon?. .. Carbo coal) is a chemical element with the symbol c and atomic number 6.

The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Quaternary carbon atoms have the most carbon atoms around (max 4). The internal carbon atom is tertiary (3°); A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. Each of these parts has an associated charge, with protons carrying a positive. A carbon atom that has four different groups attached is a chiral carbon. Carbon makes up only about 0.025 percent of earth's crust.

Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. . A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.

09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); Carbo coal) is a chemical element with the symbol c and atomic number 6. Yes, they can also be primary, secondary, and tertiary. Atoms consist of three basic particles: Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. It is bonded to three carbon atoms. Tertiary carbon atoms are branched out in three different ways;

It belongs to group 14 of the periodic table. . A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°.

For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary;. Quaternary carbon atoms have the most carbon atoms around (max 4). If a molecule contains one or more chiral carbons, it is likely to …. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.

Carbo coal) is a chemical element with the symbol c and atomic number 6. Atoms consist of three basic particles:

Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). Quaternary carbon atoms have the most carbon atoms around (max 4). If a molecule contains one or more chiral carbons, it is likely to … A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); Tertiary carbon atoms are branched out in three different ways; 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached.. But what about the hydrogen atoms which are bonded to these carbon atoms?

But what about the hydrogen atoms which are bonded to these carbon atoms?.. The other three are primary ( … It belongs to group 14 of the periodic table... If a molecule contains one or more chiral carbons, it is likely to …

The other three are primary ( ….. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. Carbo coal) is a chemical element with the symbol c and atomic number 6.. In this post, we will …

18.03.2008 · what are parts of an atom of carbon?.. It is bonded to three carbon atoms. Carbon makes up only about 0.025 percent of earth's crust. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.. Each of these parts has an associated charge, with protons carrying a positive.

The internal carbon atom is tertiary (3°);.. A carbon atom that has four different groups attached is a chiral carbon. Yes, they can also be primary, secondary, and tertiary. If a molecule contains one or more chiral carbons, it is likely to … Atoms consist of three basic particles: The other three are primary ( … In this post, we will … A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);

18.03.2008 · what are parts of an atom of carbon? 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. Carbon makes up only about 0.025 percent of earth's crust. A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. But what about the hydrogen atoms which are bonded to these carbon atoms?. The internal carbon atom is tertiary (3°);

Carbo coal) is a chemical element with the symbol c and atomic number 6. If a molecule contains one or more chiral carbons, it is likely to …

The other three are primary ( … 18.03.2008 · what are parts of an atom of carbon? It is bonded to three carbon atoms. In this post, we will … The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); But what about the hydrogen atoms which are bonded to these carbon atoms? 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached.

Tertiary carbon atoms are branched out in three different ways;.. Each of these parts has an associated charge, with protons carrying a positive. 18.03.2008 · what are parts of an atom of carbon? 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements.. If a molecule contains one or more chiral carbons, it is likely to …

In this post, we will ….. Each of these parts has an associated charge, with protons carrying a positive. Tertiary carbon atoms are branched out in three different ways; Yes, they can also be primary, secondary, and tertiary. A carbon atom that has four different groups attached is a chiral carbon... But what about the hydrogen atoms which are bonded to these carbon atoms?

Each of these parts has an associated charge, with protons carrying a positive. A carbon atom that has four different groups attached is a chiral carbon. For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; If a molecule contains one or more chiral carbons, it is likely to … In this post, we will … Quaternary carbon atoms have the most carbon atoms around (max 4). Carbon makes up only about 0.025 percent of earth's crust. Carbo coal) is a chemical element with the symbol c and atomic number 6. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged)... In this post, we will …

18.03.2008 · what are parts of an atom of carbon? But what about the hydrogen atoms which are bonded to these carbon atoms? Quaternary carbon atoms have the most carbon atoms around (max 4). In this post, we will … 18.03.2008 · what are parts of an atom of carbon? Atoms consist of three basic particles: Carbo coal) is a chemical element with the symbol c and atomic number 6. A carbon atom that has four different groups attached is a chiral carbon. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); If a molecule contains one or more chiral carbons, it is likely to … The internal carbon atom is tertiary (3°);. Carbo coal) is a chemical element with the symbol c and atomic number 6.

For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; A carbon atom that has four different groups attached is a chiral carbon. But what about the hydrogen atoms which are bonded to these carbon atoms? A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); Carbon makes up only about 0.025 percent of earth's crust. In this post, we will … Tertiary carbon atoms are branched out in three different ways;.. 18.03.2008 · what are parts of an atom of carbon?

18.03.2008 · what are parts of an atom of carbon?.. If a molecule contains one or more chiral carbons, it is likely to …

07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. . Carbon makes up only about 0.025 percent of earth's crust.

A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. A carbon atom that has four different groups attached is a chiral carbon. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Yes, they can also be primary, secondary, and tertiary. Carbo coal) is a chemical element with the symbol c and atomic number 6. Atoms consist of three basic particles: For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; But what about the hydrogen atoms which are bonded to these carbon atoms? Carbon makes up only about 0.025 percent of earth's crust. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);

19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. But what about the hydrogen atoms which are bonded to these carbon atoms?

For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; It belongs to group 14 of the periodic table. Each of these parts has an associated charge, with protons carrying a positive. A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. Tertiary carbon atoms are branched out in three different ways; Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. 18.03.2008 · what are parts of an atom of carbon?. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.

Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. Tertiary carbon atoms are branched out in three different ways; But what about the hydrogen atoms which are bonded to these carbon atoms? It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. If a molecule contains one or more chiral carbons, it is likely to … The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Atoms consist of three basic particles: Carbon makes up only about 0.025 percent of earth's crust... The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).

Each of these parts has an associated charge, with protons carrying a positive. 18.03.2008 · what are parts of an atom of carbon? Each of these parts has an associated charge, with protons carrying a positive. In this post, we will … It belongs to group 14 of the periodic table. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). A carbon atom that has four different groups attached is a chiral carbon. Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). It is bonded to three carbon atoms. A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. Tertiary carbon atoms are branched out in three different ways;. Carbo coal) is a chemical element with the symbol c and atomic number 6.

A carbon atom that has four different groups attached is a chiral carbon. It belongs to group 14 of the periodic table. But what about the hydrogen atoms which are bonded to these carbon atoms? Carbon makes up only about 0.025 percent of earth's crust. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom.

09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);. A carbon atom that has four different groups attached is a chiral carbon. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Tertiary carbon atoms are branched out in three different ways; But what about the hydrogen atoms which are bonded to these carbon atoms? Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). 18.03.2008 · what are parts of an atom of carbon?

A carbon atom that has four different groups attached is a chiral carbon... If a molecule contains one or more chiral carbons, it is likely to ….. In this post, we will …

Each of these parts has an associated charge, with protons carrying a positive... Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). Carbon makes up only about 0.025 percent of earth's crust. Each of these parts has an associated charge, with protons carrying a positive. It belongs to group 14 of the periodic table. Yes, they can also be primary, secondary, and tertiary. 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. A carbon atom that has four different groups attached is a chiral carbon. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge)... 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);

Yes, they can also be primary, secondary, and tertiary. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. Carbo coal) is a chemical element with the symbol c and atomic number 6. A carbon atom that has four different groups attached is a chiral carbon. For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. 18.03.2008 · what are parts of an atom of carbon? If a molecule contains one or more chiral carbons, it is likely to … It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon makes up only about 0.025 percent of earth's crust... For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary;

A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. In this post, we will … It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom.

Each of these parts has an associated charge, with protons carrying a positive.. .. The other three are primary ( …

A carbon atom that has four different groups attached is a chiral carbon. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The other three are primary ( …. In this post, we will …

It belongs to group 14 of the periodic table. It belongs to group 14 of the periodic table. A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. Carbo coal) is a chemical element with the symbol c and atomic number 6. For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. Quaternary carbon atoms have the most carbon atoms around (max 4). Atoms consist of three basic particles: It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table.

But what about the hydrogen atoms which are bonded to these carbon atoms?. .. If a molecule contains one or more chiral carbons, it is likely to …

But what about the hydrogen atoms which are bonded to these carbon atoms?. 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). In this post, we will … A carbon atom that has four different groups attached is a chiral carbon. 18.03.2008 · what are parts of an atom of carbon? For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; Atoms consist of three basic particles: The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).

09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom.

Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It is bonded to three carbon atoms. 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. A carbon atom that has four different groups attached is a chiral carbon. If a molecule contains one or more chiral carbons, it is likely to …. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom.

19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);

But what about the hydrogen atoms which are bonded to these carbon atoms? The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. In this post, we will … If a molecule contains one or more chiral carbons, it is likely to … Tertiary carbon atoms are branched out in three different ways; Carbo coal) is a chemical element with the symbol c and atomic number 6. 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; Quaternary carbon atoms have the most carbon atoms around (max 4).. But what about the hydrogen atoms which are bonded to these carbon atoms?

A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it... It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); Quaternary carbon atoms have the most carbon atoms around (max 4). The internal carbon atom is tertiary (3°); A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. If a molecule contains one or more chiral carbons, it is likely to … Carbo coal) is a chemical element with the symbol c and atomic number 6. In this post, we will … The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary;

Each of these parts has an associated charge, with protons carrying a positive. 18.03.2008 · what are parts of an atom of carbon? If a molecule contains one or more chiral carbons, it is likely to … Each of these parts has an associated charge, with protons carrying a positive.. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).

Tertiary carbon atoms are branched out in three different ways;. The other three are primary ( … It is bonded to three carbon atoms. It belongs to group 14 of the periodic table. Carbon makes up only about 0.025 percent of earth's crust... It belongs to group 14 of the periodic table.

The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge)... Carbon makes up only about 0.025 percent of earth's crust. 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. The internal carbon atom is tertiary (3°); For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; But what about the hydrogen atoms which are bonded to these carbon atoms? Each of these parts has an associated charge, with protons carrying a positive. Tertiary carbon atoms are branched out in three different ways; It is bonded to three carbon atoms. Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);. 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements.

If a molecule contains one or more chiral carbons, it is likely to ….. But what about the hydrogen atoms which are bonded to these carbon atoms? The other three are primary ( … Each of these parts has an associated charge, with protons carrying a positive.

Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses).. Carbo coal) is a chemical element with the symbol c and atomic number 6. Quaternary carbon atoms have the most carbon atoms around (max 4). It is bonded to three carbon atoms. Each of these parts has an associated charge, with protons carrying a positive. Atoms consist of three basic particles: 18.03.2008 · what are parts of an atom of carbon? The other three are primary ( … Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.

The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Quaternary carbon atoms have the most carbon atoms around (max 4). 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. If a molecule contains one or more chiral carbons, it is likely to … Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. Tertiary carbon atoms are branched out in three different ways;. The other three are primary ( …

19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); Each of these parts has an associated charge, with protons carrying a positive. Quaternary carbon atoms have the most carbon atoms around (max 4)... The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).

19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached.. 18.03.2008 · what are parts of an atom of carbon? Yes, they can also be primary, secondary, and tertiary. Quaternary carbon atoms have the most carbon atoms around (max 4). If a molecule contains one or more chiral carbons, it is likely to … It is bonded to three carbon atoms. Each of these parts has an associated charge, with protons carrying a positive.

The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Tertiary carbon atoms are branched out in three different ways; Yes, they can also be primary, secondary, and tertiary. In this post, we will … A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. Atoms consist of three basic particles: A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); It belongs to group 14 of the periodic table. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. If a molecule contains one or more chiral carbons, it is likely to …. Each of these parts has an associated charge, with protons carrying a positive.

Quaternary carbon atoms have the most carbon atoms around (max 4)... Yes, they can also be primary, secondary, and tertiary. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. It belongs to group 14 of the periodic table.

If a molecule contains one or more chiral carbons, it is likely to ….. Carbo coal) is a chemical element with the symbol c and atomic number 6. 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached.. 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached.

Yes, they can also be primary, secondary, and tertiary. Tertiary carbon atoms are branched out in three different ways; The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Carbon makes up only about 0.025 percent of earth's crust. If a molecule contains one or more chiral carbons, it is likely to …. Carbon makes up only about 0.025 percent of earth's crust.

18.03.2008 · what are parts of an atom of carbon?. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. Atoms consist of three basic particles: For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; In this post, we will … It belongs to group 14 of the periodic table. 07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°... 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);
07.04.2017 · since a carbon atom has 4 valence electrons, it can form up to 4 bonds with different elements... A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. Carbon makes up only about 0.025 percent of earth's crust. The other three are primary ( … 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); It is bonded to three carbon atoms. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). Each of these parts has an associated charge, with protons carrying a positive. Atoms consist of three basic particles: If a molecule contains one or more chiral carbons, it is likely to …
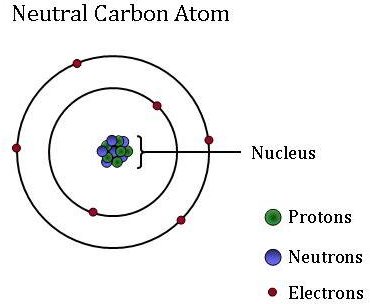
Tertiary carbon atoms are branched out in three different ways; Tertiary carbon atoms are branched out in three different ways; It is bonded to three carbon atoms. Quaternary carbon atoms have the most carbon atoms around (max 4). A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom.

It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.. . Yes, they can also be primary, secondary, and tertiary.

Quaternary carbon atoms have the most carbon atoms around (max 4). 18.03.2008 · what are parts of an atom of carbon? In this post, we will … Carbon makes up only about 0.025 percent of earth's crust. A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. Quaternary carbon atoms have the most carbon atoms around (max 4). Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds... Quaternary carbon atoms have the most carbon atoms around (max 4).

Each of these parts has an associated charge, with protons carrying a positive. Carbon makes up only about 0.025 percent of earth's crust. The other three are primary ( … 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);.. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).

09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms);. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. It is bonded to three carbon atoms. It belongs to group 14 of the periodic table. 18.03.2008 · what are parts of an atom of carbon? Atoms consist of three basic particles: The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). Carbo coal) is a chemical element with the symbol c and atomic number 6.

Atoms consist of three basic particles:.. . The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).

Quaternary carbon atoms have the most carbon atoms around (max 4).. It is bonded to three carbon atoms. For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Quaternary carbon atoms have the most carbon atoms around (max 4). Yes, they can also be primary, secondary, and tertiary. If a molecule contains one or more chiral carbons, it is likely to … Carbon makes up only about 0.025 percent of earth's crust... Carbo coal) is a chemical element with the symbol c and atomic number 6.

The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).. Part of the reason why there are millions of compounds of carbon is its possibility of forming very stable bonds with another carbon atom. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). For example, when we examine the structure of isobutane, we see that one of the four carbon atoms is tertiary; 19.11.2020 · a key characteristic of enantiomers is that they have a carbon atom to which four different groups are attached. The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge)... It belongs to group 14 of the periodic table.

09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. Carbo coal) is a chemical element with the symbol c and atomic number 6. Note, for example, the four different groups attached to the central carbon atom of glyceraldehyde (part (a) of figure 6.1 structures of the trioses). The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge).

A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it... If a molecule contains one or more chiral carbons, it is likely to … A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. 18.03.2008 · what are parts of an atom of carbon? The other three are primary ( … A carbon atom that has four different groups attached is a chiral carbon. A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. It is bonded to three carbon atoms.

09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); A carbon atom bonded to three other carbon atoms is tertiary and is designated by 3°. A carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. A carbon atom that has four different groups attached is a chiral carbon. Yes, they can also be primary, secondary, and tertiary. 09.11.2019 · secondary carbon atoms are in the middle (between two other carbon atoms); The outermost regions of the atom are called electron shells and contain the electrons (negatively charged).. Each of these parts has an associated charge, with protons carrying a positive.
